To tackle our scientific objectives, we are using multiple approaches spanning from systems to molecules and back (see pictures below). We are using and developing optical, electrophysiological, and computational tools to reveal how auditory cortical circuits are organized, built, and remodeled across development and aging. We integrate this information into computational models.
In vivo two- and three-photon imaging and holographic optogenetic stimulation
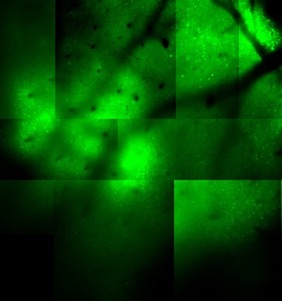
We were one of the first groups to use in vivo 2-photon imaging in auditory cortex (Bandyopadhyay et al 2010). With 2-photon imaging, we can observe the activity of thousands of neurons over large areas and volumes with single-cell resolution (see images below, every round spot is a neuron). The left image is an older “classic” image from ~2007 using synthetic dyes. The right image is from 2024 using genetically encoded indicators (imaged by Travis Babola) and the cells are color coded based on their frequency preference so you can see the large-scale tonotopy in A1 L4.
We are also the first combining in vivo 2-photon imaging with in vivo 2-photon holographic stimulation in the auditory cortex to probe auditory cortex in 3D and the neural code for sound perception (see holobrain.org and our first paper).
We use both commercial and custom 2- and 3-photon microscopes.
We complement imaging with single- and multi-electrode electrophysiological recording techniques to access areas we cannot image: deep in the brain or when we need high time resolution. We are now deploying Neuropixels 2.0 (Chen et al. 2025).

Imaging during behavior and high-throughput training
Imaging in behaving animals can show us which neurons might be important (Francis et al. 2018, Francis et al. 2021). To causally link neuronal activity, we are combining in vivo 2-photon imaging with 2-photon holographic stimulation in behaving animals to probe the neural code for sound perception (see holobrain.org).
Ultimately the brain controls behavior, and conversely learning changes the brain. We investigate this interaction by training animals and relating their performance and learning to the activity of specific cortical circuits. We develop automatic systems to efficiently train and test large groups of mice (Francis et al. 2017, Francis et al. 2019, Wang et al. 2024).
Circuit mapping in vitro
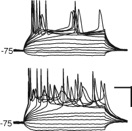
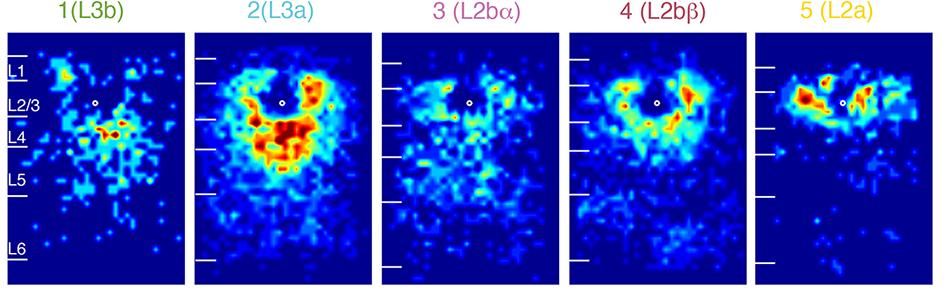
We use brain slices to study and dissect mesoscale circuits using patch-clamp and imaging techniques coupled with laser-scanning photostimulation (LSPS) (i.e. Zhao et al. 2009, Viswanathan et al. 2012. Meng et al. 2017, Meng et al. 2021 etc). With LSPS we activate presynaptic neurons and then observe the responses with whole cell patch clamp recordings. By stimulating many (up to 1000) target sites we can assemble a high resolution map of functional connectivity or functional “wiring diagram”. By comparing these wiring diagrams between animals of different ages to animals that had different experiences we can identify how for example experience shapes brain circuits.
Molecules and pathways
We augment physiological studies by amatomical, molecular, and histological methods (In situ hybridizations, qPCR, immunohistochemistry, anterograde and retrograde tracers) to get a full picture of the brain.

